Response of schizophrenia patients with different intestinal flora distribution to olanzapine
-
摘要:
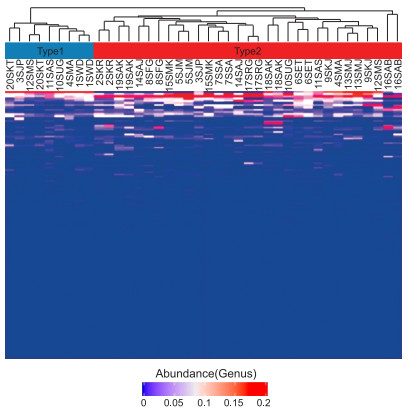
目的 探究首诊精神分裂症患者的肠道菌群分布以及不同肠道菌群分布患者对于奥氮平的反应性。 方法 选择2020年3月—2021年1月于衢州市第三人民医院精神科首次诊断和治疗的精神分裂症患者40例作为研究对象,采用16S rRNA测序以及基因数据库检测其肠道菌群分布,根据结果进行分组。采用奥氮平规律治疗8周,比较不同菌群分布的精神分裂症患者治疗效果、阳性与阴性症状量表(PANSS)评分、个人与社会表现量表(PSP)评分的差异。 结果 40例患者中,1型肠道菌群分布共9例,以普雷沃菌属为主;2型共31例,以拟杆菌属、布劳特氏菌属、粪厌氧棒杆菌、孪生菌为主。治疗8周后,1型菌群分布患者的治疗显效率、有效率和无效率分别为55.56%、44.44%、0.00%,2型菌群上述指标分别为19.35%、54.85%、25.81%,差异有统计学意义(Z=2.531,P < 0.05)。1型肠道菌群分布的患者PANSS评分低于2型(t=4.261,P < 0.05),PSP评分高于2型(t=5.921,P < 0.05)。 结论 肠道菌群以普雷沃菌属为主的精神分裂症患者对奥氮平的反应性更佳,更有利于改善症状和个人与社会表现,根据肠道菌群判断患者预后成为一种潜在的方式。 Abstract:Objective To explore the distribution of intestinal flora in patients with schizophrenia and the response of patients with different intestinal flora to olanzapine. Methods From March 2020 to January 2021, 40 schizophrenic patients who were diagnosed and treated in our hospital for the first time were selected as the research objects. 16S rRNA sequencing and gene database were used to detect the distribution of intestinal flora. According to the results, the patients were divided into groups and treated with olanzapine for 8 weeks. The scores of the positive and negative syndrome scale (PANSS), personal and social performance scale (PSP) and Wisconsin Card Sorting Test were compared. Results In 40 patients, 9 cases had type 1 flora (mainly Prevotella), and 31 cases had type 2 flora (mainly Bacteroides, Brucella, Anaerostipes and Eggerthella). After 8 weeks of treatment, the significant efficiency, effective rate and ineffective rate of treatment of patients with type 1 flora distribution were 55.56%, 44.44% and 0.00%, respectively, and those of patients with type 2 flora distribution were 19.35%, 54.85% and 25.81%, respectively. The difference was statistically significant (Z=2.531, P < 0.05). The PANSS score of patients with type 1 intestinal flora distribution was lower than that of patients with type 2 intestinal flora distribution (t=4.261, P < 0.05), and the PSP score was higher in the former than in the latter (t=5.921, P < 0.05). Conclusion Schizophrenic patients with type 1 intestinal flora distribution have better response to olanzapine, which is more conducive to improving symptoms and personal and social performance. Therefore, the prognosis of patients can be determined according to their intestinal flora. -
Key words:
- Schizophrenia /
- Intestinal flora /
- Olanzapine /
- Drug reactivity /
- Curative effect
-
表 1 不同类型肠道菌群分布的相对富集度比较
肠道菌属 相对丰度(%) FDR校正P值 1型 2型 普雷沃菌属 45.50(42.50~49.50) 0.75(0.50~1.00) < 0.001 拟杆菌属 7.60(6.00~9.20) 25.50(14.83~30.62) < 0.001 布劳特氏菌属 2.56(1.54~4.73) 6.05(5.50~7.55) 0.008 粪厌氧棒杆菌 0.09(0.02~0.18) 0.19(0.11~0.25) 0.042 孪生菌 0.03(0.01~0.05) 0.12(0.08~0.17) 0.032 表 2 2组精神分裂症患者疗效比较[例(%)]
菌属 例数 显效 有效 无效 1型 9 5(55.56) 4(44.44) 0(0.00) 2型 31 6(19.35) 17(54.84) 8(25.81) 注:2组疗效比较,Z=2.531,P=0.019。 表 3 2组精神分裂症患者治疗前后PANSS评分及PSP评分比较(x ±s,分)
菌属 例数 PANSS PSP 治疗前 治疗8周后 治疗前 治疗8周后 1型 9 76.58±7.68 38.36±4.30 45.47±3.47 65.48±6.58 2型 31 77.04±7.47 45.85±4.73 45.78±3.50 52.38±5.63 t值 0.162 4.261 0.234 5.921 P值 0.872 < 0.001 0.816 < 0.001 -
[1] 赵星梅, 王喜苹, 周火祥, 等. 基于高通量测序的精神分裂症患者肠道菌群多样性[J]. 中国微生态学杂志, 2019, 31(1): 1-7. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGWS201901001.htm [2] 黄侠, 卓敏, 李时佳, 等. 精神分裂症患者肠道菌群结构特征初步观察与分析[J]. 中国神经精神疾病杂志, 2019, 45(7): 401-406. doi: 10.3969/j.issn.1002-0152.2019.07.004 [3] XU R H, WU B B, LIANG J W, et al. Altered gut microbiota and mucosal immunity in patients with schizophrenia[J]. Brain Behav Immun, 2020(85): 120-127. http://www.sciencedirect.com/science/article/pii/S0889159119300807 [4] GUBERT C, KONG G, UZUNGIL V, et al. Microbiome profiling reveals gut dysbiosis in the metabotropic glutamate receptor 5 knockout mouse model of schizophrenia[J]. Front Cell Dev Biol, 2020(8): 582320. http://www.researchgate.net/publication/346477831_Microbiome_Profiling_Reveals_Gut_Dysbiosis_in_the_Metabotropic_Glutamate_Receptor_5_Knockout_Mouse_Model_of_Schizophrenia [5] LAINE A, ANTTILA M, HIRVONEN H, et al. Feasibility of a web-based psychoeducation course and experiences of caregivers living with a person with schizophrenia spectrum disorder: Mixed methods study[J]. J Med Internet Res, 2021, 23(4): e25480. doi: 10.2196/25480 [6] MANOSSO L M, LIN J, CARLESSI A S, et al. Sex-related patterns of the gut-microbiota-brain axis in the neuropsychiatric conditions[J]. Brain Res Bull, 2021(171): 196-208. [7] ALMEIDA C, OLIVEIRA R, SOARES R, et al. Influence of gut microbiota dysbiosis on brain function: A systematic review[J]. Porto Biomed J, 2020, 5(2): 1-8. doi: 10.1097/j.pbj.0000000000000059 [8] SELTEN J P, VAN DER VEN E, RUTTEN B P, et al. The social defeat hypothesis of schizophrenia: An update[J]. Schizophr Bull, 2013, 39(6): 1180-1186. doi: 10.1093/schbul/sbt134 [9] LI Y, ZHANG L, ZHANG Y, et al. A random forest model for predicting social functional improvement in Chinese patients with schizophrenia after 3 months of atypical antipsychotic monopharmacy: A cohort study[J]. Neuropsychiatr Dis Treat, 2021(17): 847-857. http://www.researchgate.net/publication/350164037_A_Random_Forest_Model_for_Predicting_Social_Functional_Improvement_in_Chinese_Patients_with_Schizophrenia_After_3_Months_of_Atypical_Antipsychotic_Monopharmacy_A_Cohort_Study [10] LEE Y, KIM Y K. Understanding the connection between the gut-brain axis and stress/anxiety disorders[J]. Curr Psychiatry Rep, 2021, 23(5): 22. doi: 10.1007/s11920-021-01235-x [11] KIM Y K, SHIN C. The microbiota-gut-brain axis in neuropsychiatric disorders: Pathophysiological mechanisms and novel treatments[J]. Curr Neuropharmacol, 2018, 16(5): 559-573. doi: 10.2174/1570159X15666170915141036 [12] 王彩侠, 易正辉. 肠道菌群与精神分裂症关系的研究现状及进展[J]. 中国医师杂志, 2020, 22(9): 1435-1438. doi: 10.3760/cma.j.cn431274-20190818-00961 [13] 张帆, 方星, 徐美玲, 等. 帕金森病患者肠道普氏菌改变及其预测价值[J]. 医学研究生学报, 2020, 33(5): 482-486. https://www.cnki.com.cn/Article/CJFDTOTAL-JLYB202005008.htm [14] 张言武, 白丽君, 程强, 等. 精神分裂症发作期与缓解期肠道菌群高通量测序分析[J]. 中国神经精神疾病杂志, 2018, 44(12): 705-709. doi: 10.3969/j.issn.1002-0152.2018.12.001 [15] 申变红, 陶云海, 朱春燕. 肠道菌群比例在精神分裂症发病中的作用及其与炎症因子的关系[J]. 中华全科医学, 2018, 16(2): 276-278. https://www.cnki.com.cn/Article/CJFDTOTAL-SYQY201802033.htm [16] CHEN X C, XU J J, WANG H R, et al. Profiling the differences of gut microbial structure between schizophrenia patients with and without violent behaviors based on 16S rRNA gene sequencing[J]. Int J Legal Med, 2021, 135(1): 131-141. doi: 10.1007/s00414-020-02439-1 [17] 潘忠德, 鞠培俊, 朱翠珍. 精神分裂症暴力和攻击行为的生物学研究进展[J]. 神经疾病与精神卫生, 2020, 20(12): 837-842. doi: 10.3969/j.issn.1009-6574.2020.12.001 [18] SONNWEBER M, LAU S, KIRCHEBNER J. Violent and non-violent offending in patients with schizophrenia: Exploring influences and differences via machine learning[J]. Compr Psychiatry, 2021(107): 152238. http://www.sciencedirect.com/science/article/pii/S0010440X2100016X [19] 李自辉, 庞牧, 林佩锜, 等. 基于16S rRNA技术分析柴胡龙骨牡蛎汤对精神分裂症大鼠肠道菌群多样性的影响[J]. 中国实验方剂学杂志, 2019, 25(13): 1-8. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201913001.htm [20] 李艳, 项丽玲, 郭晖, 等. 基于"心与小肠相表里"的肠道菌群对中枢神经系统的影响及中药干预[J]. 中草药, 2019, 50(6): 1493-1498. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201906035.htm -





 下载:
下载: