Effect and mechanism of low ENPP1 expression on the epithelial-mesenchymal transition of oral squamous cell carcinoma cell line
-
摘要:
目的 探讨外核苷酸焦磷酸酶/磷酸二酯酶1(ENPP1)低表达对口腔鳞状细胞癌(简称口腔鳞癌)细胞上皮间质转化的作用。 方法 取正常人口腔黏膜上皮细胞系、人口腔鳞癌细胞系CAL-27进行培养、传代,检测ENPP1 mRNA表达。取对数生长期CAL-27细胞进行转染,分为空白组(未处理)、对照组(转染NC-siRNA质粒)及转染组(转染ENPP1-siRNA),检测ENPP1 mRNA、蛋白表达。取对数生长期CAL-27细胞进行转染,分为A组(未转染)、B组(转染NC-siRNA质粒)、C组(转染ENPP1-siRNA质粒+MAPK激动剂)、D组(转染ENPP1-siRNA质粒)、E组(MAPK激动剂),检测CAL-27细胞增殖活性、侵袭细胞数、迁移能力及磷酸化丝裂原活化蛋白激酶(p-MAPK)、磷酸化胞外信号调节蛋白激酶1/2(p-ERK1/2)蛋白表达。 结果 各时段吸光度(A)值,D组<C组<A组、B组<E组(均P<0.05);A组、B组、C组、D组、E组侵袭细胞数分别为(85.23±2.22)个/视野、(85.41±1.59)个/视野、(53.67±2.10)个/视野、(37.39±2.06)个/视野、(111.08±5.20)个/视野,D组<C组<A组、B组<E组(均P<0.05);划痕宽度百分比,E组<A组、B组<C组<D组(均P<0.05);p-MAPK、p-ERK1、p-ERK2蛋白相对表达量,D组<C组<A组、B组<E组(均P<0.05)。 结论 ENPP1低表达可抑制口腔鳞癌细胞上皮间质转化,机制可能与抑制MAPK信号通路有关。 -
关键词:
- 口腔鳞状细胞癌 /
- 上皮间质转化 /
- 核苷酸焦磷酸酶/磷酸二酯酶1
Abstract:Objective To investigate the effect of low expression ectonucleotide pyrophosphatase/phosphodiesterase 1 (ENPP1) on the epithelial-mesenchymal transition (EMT) in oral squamous cell carcinoma. Methods The normal human oral mucosal epithelial cell line and human oral squamous cell carcinoma CAL-27 cells were cultured and subcultured, the expression of ENPP1 mRNA was detected. CAL-27 cells in logarithmic growth stage were transfected and divided into blank group (without treatment), control group (transfected with NC-siRNA plasmid) and transfection group (transfected with ENPP1-siRNA). The expression of ENPP1 mRNA and protein were detected. CAL-27 cells in logarithmic growth stage were transfected and divided into group A (not transfected), group B (transfected with NC-siRNA plasmid) and group C (transfected with ENPP1-siRNA plasmid + MAPK agonist), group D (transfected with ENPP1-siRNA plasmid), group E (MAPK agonist). The proliferation activity, number of invasive cells, migration ability and protein expression of phosphorylated-mitogen activated protein kinase (p-MAPK) and phosphorylated-extracellular regulated kinase 1/2 (p-ERK1/2) were detected. Results Absorbance (A) of each period, group D < group C < group A, group B < group E (all P < 0.05). The number of invasive cells in five groups were (85.23±2.22) cells/field, (85.41±1.59) cells/field, (53.67±2.10) cells/field, (37.39±2.06) cells/field and (111.08±5.20) cells/field cells/field, respectively, group D < group C < group A, group B < group E (all P < 0.05). The percentage of scratch width: group E < group A, group B < group C < group D (all P < 0.05). The relative expression levels of p-MAPK, p-ERK1 and p-ERK2 proteins: group D < group C < group A, group B < group E (all P < 0.05). Conclusion Low expression of ENPP1 can inhibit EMT, the mechanism may be related to the inhibition of MAPK signaling pathway. -
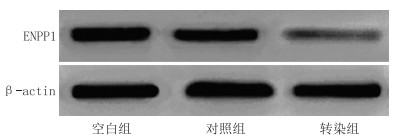
表 1 各组细胞ENPP1 mRNA、蛋白相对表达量比较(x±s)
Table 1. Comparison of the relative expression levels of ENPP1 mRNA and protein in each group of cells (x±s)
组别 n ENPP1 mRNA ENPP1蛋白 空白组 5 0.64±0.02 0.73±0.03 对照组 5 0.63±0.02 0.72±0.03 转染组 5 0.31±0.02ab 0.24±0.02ab F值 328.086 524.770 P值 < 0.001 <0.001 注:与空白组比较,aP<0.05;与对照组比较,bP<0.05。 表 2 各组细胞MTT实验A值比较(x±s,%)
Table 2. Comparison of A value in MTT experiment of cells in each group(x±s)
组别 n 24 h 48 h 72 h A组 5 30.68±1.65 39.76±1.69e 52.22±3.61ef B组 5 30.35±1.35 39.86±1.74e 51.49±2.51ef C组 5 19.09±1.02ab 29.62±1.59abe 40.42±2.25abef D组 5 8.33±0.70abc 14.70±1.44abce 21.04±1.86abcef E组 5 38.94±1.93abcd 54.91±1.60abcde 65.55±1.79abcdef F值 289.917 337.243 178.047 P值 <0.001 <0.001 <0.001 注:与A组比较,aP<0.05;与B组比较,bP<0.05;与C组比较,cP<0.05;与D组比较,dP<0.05;与本组培养24 h时比较,eP<0.05;与本组培养48 h时比较,fP<0.05。 表 3 各组CAL-27细胞侵袭细胞数比较(x±s,个/视野)
Table 3. Comparison of the number of invasive cells of CAL-27 cells in each group (x±s, pcs/field of view)
组别 n 侵袭细胞数 A组 5 85.23±2.22 B组 5 84.51±1.59 C组 5 53.67±2.10ab D组 5 37.39±2.06abc E组 5 111.08±5.20abcd F值 390.643 P值 <0.001 注:与A组比较,aP<0.05;与B组比较,bP<0.05;与C组比较,cP<0.05;与D组比较,dP<0.05。 表 4 各组CAL-27细胞划痕宽度百分比比较(x±s,%)
Table 4. Comparison of the percentage of scratch width of CAL-27 cells in each group (x±s, %)
组别 n 划痕宽度 A组 5 44.73±1.78 B组 5 44.05±2.09 C组 5 57.99±1.17ab D组 5 95.59±2.15abc E组 5 31.08±2.21abcd F值 666.858 P值 <0.001 注:与A组比较,aP<0.05;与B组比较,bP<0.05;与C组比较,cP<0.05;与D组比较,dP<0.05。 表 5 各组细胞p-MAPK、p-ERK1、p-ERK2蛋白相对表达量比较(x±s)
Table 5. Comparison of the relative expression levels of p-MAPK, p-ERK1 and p-ERK2 proteins in each group of cells (x±s)
组别 n p-MAPK p-ERK1 p-ERK2 A组 5 0.37±0.02 0.66±0.03 0.52±0.02 B组 5 0.36±0.02 0.65±0.02 0.52±0.02 C组 5 0.27±0.01ab 0.32±0.02ab 0.33±0.01ab D组 5 0.12±0.01abc 0.14±0.01abc 0.15±0.01abc E组 5 0.75±0.02abcd 1.01±0.02abcd 1.13±0.05abcd F值 845.950 1 042.375 844.352 P值 <0.001 <0.001 <0.001 注:与A组比较,aP<0.05;与B组比较,bP<0.05;与C组比较,cP<0.05;与D组比较,dP<0.05。 -
[1] 刘璐璐, 李秀川, 袁冯, 等. 加速康复外科理念在游离皮瓣修复口腔癌患者中的应用[J]. 中华全科医学, 2020, 18(10): 1774-1778. doi: 10.16766/j.cnki.issn.1674-4152.001616LIU L L, LI X C, YUAN F, et al. Application of the concept of enhanced recovery after surgery in free flap repair of oral cancer patients[J]. Chinese Journal of General Practice, 2020, 18(10): 1774-1778. doi: 10.16766/j.cnki.issn.1674-4152.001616 [2] SHETTY S S, SHARMA M, FONSECA F P, et al. Signaling pathways promoting epithelial mesenchymal transition in oral submucous fibrosis and oral squamous cell carcinoma[J]. Jpn Dent Sci Rev, 2020, 56(1): 97-108. doi: 10.1016/j.jdsr.2020.07.002 [3] LUO Y D, DING X, DU H M, et al. FOXM1 is a novel predictor of recurrence in patients with oral squamous cell carcinoma associated with an increase in epithelial-mesenchymal transition[J]. Mol Med Rep Vol, 2019, 19(5): 4101-4108. [4] 赵伟萍, 李合芳, 马妍敏, 等. 苦参碱注射液对中晚期口腔癌患者炎症、凋亡及侵袭转移相关因子表达的影响[J]. 河北医药, 2020, 42(3): 378-381. doi: 10.3969/j.issn.1002-7386.2020.03.013ZHAO W P, LI H F, MA Y M, et al. Effects of matrine injection on expressions of inflammation, apoptosis and invasion and metastasis-related factors in patients with advanced oral cancer[J]. Hebei Medical Journal, 2020, 42(3): 378-381. doi: 10.3969/j.issn.1002-7386.2020.03.013 [5] HU M, GUO W, LIAO Y, et al. Dysregulated ENPP1 increases the malignancy of human lung cancer by inducing epithelial-mesenchymal transition phenotypes and stem cell features[J]. Am J Cancer Res, 2019, 9(1): 134-144. [6] KAWECKI, NEDEVA I R, ILOYA J, et al. Mouth cancer awareness in general population: Results from grampian region of scotland, united kingdom[J]. J Oral Maxillofac Res, 2019, 10(2): e3-e5. [7] GHAFOURI F S, GHOLIPOUR M, TAHERI M, et al. MicroRNA profile in the squamous cell carcinoma: Prognostic and diagnostic roles[J]. Heli, 2020, 6(11): e05436. DOI: 10.1016/j.heliyon.2020.e05436. [8] LEE C H. Reversal of epithelial-mesenchymal transition by natural anti-inflammatory and pro-resolving lipids[J]. 1841. DOI: 10.3390/cancers11121841. [9] WILLIAMS E D, GAO D, REDFERN A, et al. Controversies around epithelial-mesenchymal plasticity in cancer metastasiss[J]. Nat Rev Cancer, 2019, 19(12): 716-732. doi: 10.1038/s41568-019-0213-x [10] CURTIS V F, CARTWRIGHT I M, LEE J S, et al. Neutrophils as sources of dinucleotide polyphosphates and metabolism by epithelial ENPP1 to influence barrier function via adenosine signaling[J]. Mol Biol Cell, 2018, 29(22): 2687-2699. doi: 10.1091/mbc.E18-06-0377 [11] STABACH P R, ZIMMERMAN K, ADAME A, et al. Improving the pharmacodynamics and in vivo activity of ENPP1-Fc through protein and glycosylation engineering[J]. Clin Transl Sci, 2021, 14(1): 362-372. doi: 10.1111/cts.12887 [12] OHEIM R, ZIMMERMAN K, MAULDING N D, et al. Human heterozygous ENPP1 deficiency is associated with early onset osteoporosis, a phenotype recapitulated in a mouse model of Enpp1 deficiency[J]. J Bone Miner Res, 2020, 35(3): 528-539. doi: 10.1002/jbmr.3911 [13] WANG H, YE F, ZHOU C, et al. High expression of ENPP1 in high-grade serous ovarian carcinoma predicts poor prognosis and as a molecular therapy target[J]. PLoS One, 2021, 16(2): e0245733. DOI: 10.1371/journal.pone.0245733. [14] ONYEDIBE K I, WANG M, SINTIM H O. ENPP1, an old enzyme with New functions, and small molecule inhibitors-A STING in the tale of ENPP1[J]. Mole, 2019, 24(22): 4192-4193. doi: 10.3390/molecules24224192 [15] KOTWAL A, FERRER A, KUMAR R, et al. Clinical and biochemical phenotypes in a family with ENPP1 mutations[J]. J Bone Miner Res, 2020, 35(4): 662-670. doi: 10.1002/jbmr.3938 [16] 马超, 张韬, 林润台, 等. ENPP1诱导肿瘤干性和上皮间质转化促进口腔鳞癌的发展[J]. 现代口腔医学杂志, 2020, 34(3): 129-132. https://www.cnki.com.cn/Article/CJFDTOTAL-XDKY202003003.htmMA C, ZHANG T, LIN R T, et al. ENPP1 induces stemness and epithelial-mesenchymal transition in oral squamous cell carcinoma[J]. Journal of contemporary stomatology, 2020, 34(3): 129-132. https://www.cnki.com.cn/Article/CJFDTOTAL-XDKY202003003.htm [17] BRAICU C, BUSE M, BUSUIOC C, et al. A comprehensive review on MAPK: A promising therapeutic target in cancer[J]. Cancers, 2019, 11(10): 1618-1619. doi: 10.3390/cancers11101618 [18] DUSHANE J K, MAGINNIS M S. Human DNA virus exploitation of the MAPK-ERK cascade[J]. Int J Mol Sci, 2019, 20(14): 3427-3428. doi: 10.3390/ijms20143427 [19] 林镇海, 闫士灿, 张洁筠, 等. MST4通过激活MAPK-ERK信号通路调节炎症因子释放促进肝癌的侵袭和转移的机制研究[J]. 中国癌症杂志, 2017, 27(9): 681-686. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGAZ201709001.htmLIN Z H, YAN S C, ZHANG J Y, et al. MST4 upregulates secretion of inflammatory factors via activation of MAPK-ERK signaling pathway and promotes invasion and metastasis of hepatocellular carcinoma[J]. China Oncology, 2017, 27(9): 681-686. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGAZ201709001.htm [20] 朱晨笛, 郭慕真, 蔡倩, 等. MAPK信号通路在百草枯诱导上皮-间充质改变中的作用[J]. 中华劳动卫生职业病杂志, 2018, 36(8): 561-567. doi: 10.3760/cma.j.issn.1001-9391.2018.08.001ZHU C D, GUO M Z, CAI Q, et al. Role of MAPK signaling pathway in epithelial-mesenchymal transition of type Ⅱ alveolar epithelial cells induced by Paraquat[J]. Chinese Journal of Industrial Hygiene and Occupational Diseases, 2018, 36(8): 561-567. doi: 10.3760/cma.j.issn.1001-9391.2018.08.001 [21] CHEN K, LIU M X, MAK C S, et al. Methylation-associated silencing of miR-193a-3p promotes ovarian cancer aggressiveness by targeting GRB7 and MAPK/ERK pathways[J]. Theranostics, 2018, 8(2): 423-436. doi: 10.7150/thno.22377 -





 下载:
下载: