Analysis of the disease characteristics and differential diagnostic factors of patients with Helicobacter pylori infection-related gastritis and autoimmune gastritis
-
摘要:
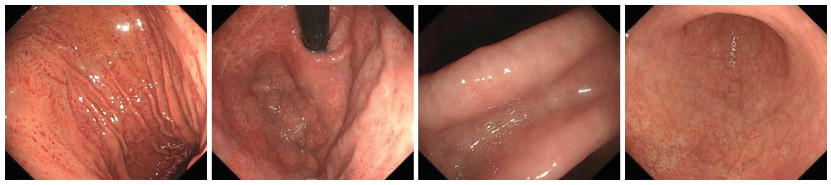
目的 分析幽门螺旋杆菌感染相关胃炎与自身免疫性胃炎(AIG)患者病情特征,并探究影响鉴别诊断的因素,为早期识别胃炎类型提供理论依据。 方法 选取2022年9月—2023年12月于宁波大学附属阳明医院治疗的49例幽门螺旋杆菌(Hp)感染相关性胃炎患者为Hp组,选取同期49例自身免疫性胃炎患者为AIG组。收集2组患者的临床资料,并对患者病情特征和鉴别诊断因素进行回顾性分析及总结。 结果 2组患者胃轻瘫主要症状指数(GCSI)、抗内因子抗体(IFA)阳性、胃蛋白酶原Ⅰ(PGⅠ)、胃蛋白酶原Ⅱ(PGⅡ)、PGⅠ/PGⅡ值、胃泌素-17(G-17)比较,差异均有统计学意义(P < 0.05)。Hp相关性胃炎胃镜下可见胃窦黏膜红白相间,以白为主,黏膜下血管透见。AIG胃镜下可见胃底、胃体黏膜变薄,红白相间,以白为主,黏膜下血管透见,可见增生性息肉,而胃窦部相对正常。Hp相关性胃炎组织病理学表现为固有层浅表单核炎性细胞和胃腺颈部中性粒细胞的弥漫性浸润,可见肠型上皮细胞取代泌酸腺黏膜中的上皮细胞。AIG组织病理学表现为固有层中基底层为主的慢性炎性细胞浸润,可见腺上皮的假幽门化生或胰腺腺泡细胞化生。多因素logistic分析显示,PGⅠ/PGⅡ值(OR=0.278,P < 0.001)、G-17(OR=1.268,P=0.005)均为影响Hp相关性胃炎与AIG鉴别诊断的因素。 结论 G-17、PGⅠ/PGⅡ值和组织病理学可用于鉴别Hp相关性胃炎和AIG,以提高诊断的准确性。 -
关键词:
- 幽门螺旋杆菌相关性胃炎 /
- 自身免疫性胃炎 /
- 诊断 /
- 胃镜
Abstract:Objective To analyze the disease characteristics of patients with Helicobacter pylori (Hp) infection-related gastritis and autoimmune gastritis (AIG), and to explore the factors influencing differential diagnosis, so as to provide a theoretical basis for the early identification of gastritis types. Methods A total of 49 patients diagnosed with Helicobacter pylori infection-associated gastritis who received treatment at Yangming Hospital Affiliated to Ningbo University from September 2022 to December 2023 were selected for the Hp group, and 49 patients with autoimmune gastritis during the same period were selected for the AIG group. The clinical data of the two groups of patients were collected. A retrospective analysis and summary were conducted on the disease characteristics and differential diagnostic factors. Results There were statistically significant differences in gastroparesis cardinal symptom index (GCSI), anti-intrinsic factor antibodies (IFA) positivity, pepsinogen Ⅰ (PG Ⅰ), pepsinogen Ⅱ (PG Ⅱ), PG Ⅰ/PG Ⅱ ratio, and gastrin-17 (G-17) between the two groups of patients (P < 0.05). During gastroscopy, the antral mucosa of HP-related gastritis showed a mixture of red and white, with white being dominant, and submucosal blood vessels were visible. Under AIG gastroscopy, it can be seen that the mucosa in the fundus and body of the stomach is thinning, with alternating red and white regions, primarily white. The presence of submucosal blood vessels are visible, and hyperplastic polyps can be seen, while the antrum of the stomach was relatively normal. The histopathological manifestations of HP-related gastritis were characterized by diffuse infiltration of superficial mononuclear inflammatory cells in the lamina propria and neutrophils in the neck of the gastric glands. Intestinal-type epithelial cells have been observed to replace the epithelial cells in the mucosa of the acid-secreting glands. The histopathological manifestation of AIG was chronic inflammatory cell infiltration, primarily in the basal layer of the lamina propria, with pseudopyloric metaplasia of the glandular epithelium or pancreatic acinar cell metaplasia being visible. Multivariate logistic analysis demonstrated that the PG Ⅰ/PG Ⅱ ratio (OR=0.278, P < 0.001) and G-17 (OR=1.268, P=0.005) were the factors influencing the differential diagnosis between HP-related gastritis and AIG. Conclusion The G17, PG Ⅰ/PG Ⅱ ratio and histopathology can be used to distinguish Hp-related gastritis from AIG, thus improving the accuracy of diagnosis. -
Key words:
- Helicobacter pylori-associated gastritis /
- Autoimmune gastritis /
- Diagnosis /
- Gastroscopy
-
表 1 2组胃炎患者临床资料比较
Table 1. Comparison of clinical data of the two groups of patients with gastritis
项目 HP组(n=49) AIG组(n=49) 统计量 P值 年龄(x±s, 岁) 56.38±3.98 55.46±4.67 1.050a 0.297 性别[例(%)] 0.172b 0.678 男性 29(59.18) 31(63.27) 女性 20(40.82) 18(36.73) BMI(x±s) 20.17±2.59 19.26±3.04 1.595a 0.114 吸烟史[例(%)] 27(55.10) 29(59.18) 0.167b 0.683 饮酒史[例(%)] 21(42.86) 24(48.98) 0.370b 0.543 合并疾病[例(%)] 高血压 11(22.45) 13(26.53) 0.221b 0.638 糖尿病 14(28.57) 17(34.69) 0.425b 0.515 冠心病 10(20.41) 9(18.37) 0.065b 0.798 贫血史[例(%)] 18(36.73) 26(53.06) 2.640b 0.104 桥本甲状腺炎史[例(%)] 10(20.41) 18(36.73) 3.200b 0.074 注:a为t值,b为χ2值。 表 2 2组胃炎患者GCSI评分及实验室指标比较
Table 2. Comparison of GCSI scores and laboratory indicators between the two groups of patients with gastritis
组别 例数 GCSI评分(x±s, 分) IFA阳性[例(%)] PCA阳性[例(%)] PGⅠ (x±s, μg/L) PGⅡ (x±s, μg/L) PGⅠ/PGⅡ (x±s) G-17 (x±s, pmol/L) VB12 (x±s, pmol/L) 血红蛋白(x±s, g/L) HP组 49 7.28±1.95 10(20.41) 9(18.37) 70.91±19.45 8.84±2.35 10.35±2.67 14.69±3.51 331.57±69.23 129.76±26.54 AIG组 49 8.36±2.01 19(38.78) 17(34.69) 63.95±12.47 10.24±2.78 6.11±1.49 25.52±7.26 319.98±73.46 121.33±29.68 统计量 2.700a 3.967b 3.350b 2.109a 2.692a 9.707a 9.401a 0.804a 1.482a P值 0.008 0.046 0.067 0.038 0.008 < 0.001 < 0.001 0.424 0.142 注:a为t值,b为χ2值。 表 3 各变量赋值情况
Table 3. Assignment of each variable
变量 赋值方法 GCSI评分 连续变量,以实际值赋值 IFA阳性 有=1,无=0 PGⅠ 连续变量,以实际值赋值 PGⅡ 连续变量,以实际值赋值 PGⅠ/PGⅡ 连续变量,以实际值赋值 G-17 连续变量,以实际值赋值 胃炎类型 AIG组=1,Hp组=0 表 4 影响幽门螺旋杆菌感染相关胃炎与AIG鉴别诊断的单因素logistic回归分析
Table 4. Univariate logistic regression analysis on the differential diagnosis between helicobacter pylori infection-related gastritis and AIG
变量 B SE Waldχ2 P值 OR值 95% CI GCSI评分 0.194 0.100 3.750 0.053 1.214 0.998~1.477 IFA阳性 0.859 0.476 3.263 0.071 2.361 0.930~5.997 PGⅠ -0.035 0.014 5.840 0.016 0.966 0.939~0.993 PGⅡ 0.110 0.071 2.397 0.122 1.117 0.971~1.284 PGⅠ/PGⅡ值 -1.328 0.297 19.995 < 0.001 0.265 0.148~0.474 G-17 0.337 0.070 23.175 < 0.001 1.401 1.221~1.607 表 5 影响幽门螺旋杆菌感染相关胃炎与AIG鉴别诊断的多因素logistic回归分析
Table 5. Multivariate logistic regression analysis on the differential diagnosis between helicobacter pylori infection-related gastritis and AIG
变量 B SE Waldχ2 P值 OR值 95% CI PGⅠ -0.068 0.044 2.329 0.127 0.935 0.857~1.019 PGⅠ/PGⅡ -1.281 0.351 13.315 < 0.001 0.278 0.140~0.553 G-17 0.238 0.085 7.758 0.005 1.268 1.073~1.499 -
[1] KAMETAKA D, IWAMURO M, TAKAHASHI T, et al. Characterization of gastric tissue-resident T cells in autoimmune and Helicobacter pylori-associated gastritis[J]. Curr Issues Mol Biol, 2022, 44(6): 2443-2452. doi: 10.3390/cimb44060167 [2] SHUKLA G T, YADAV S, SHUKLA A, et al. Histopathological features of chronic gastritis and its association with Helicobacter pylori infection[J]. Korean J Gastroenterol, 2024, 84(4): 153-159. doi: 10.4166/kjg.2024.063 [3] LENTI M V, RUGGE M, LAHNER E, et al. Autoimmune gastritis[J]. Nat Rev Dis Primers, 2020, 6(1): 57. DOI: 10.1038/s41572-020-0198-5. [4] ALVAREZ-ALDANA A, IKHIMIUKOR O O, GUACA-GONZÁLEZ Y M, et al. Genomic insights into the antimicrobial resistance and virulence of Helicobacter pylori isolates from gastritis patients in Pereira, Colombia[J]. BMC Genomics, 2024, 25(1): 843. DOI: 10.1186/s12864-024-10749-6. [5] YU Y F, TONG K K, SHANGGUAN X L, et al. Research status and hotspots of autoimmune gastritis: a bibliometric analysis[J]. World J Gastroenterol, 2023, 29(42): 5781-5799. doi: 10.3748/wjg.v29.i42.5781 [6] BUTT J, LEHTINEN M, ÖHMAN H, et al. Association of Helicobacter pylori and autoimmune gastritis with stomach cancer in a cohort of young finnish women[J]. Gastroenterology, 2022, 163(1): 305-307. doi: 10.1053/j.gastro.2022.03.012 [7] 房静远, 杜奕奇, 刘文忠, 等. 中国慢性胃炎诊治指南(2022年, 上海)[J]. 胃肠病学, 2022, 27(4): 193-224.FANG J Y, DU Y Q, LIU W Z, et al. Chinese guidelines for diagnosis and treatment of chronic gastritis (2022, Shanghai)[J]. Chin J Gastroenter, 2022, 27(4): 193-224. [8] 胡海一, 陈楚岩, 王巍. 自身免疫性胃炎患者胃排空延迟的回顾性研究[J]. 临床和实验医学杂志, 2025, 24(9): 897-901.HU H Y, CHEN C Y, WANG W. A retrospective study on delayed gastric emptying in patients with autoimmune gastritis[J]. Journal of Clinical and Experimental Medicine, 2020, 24(9): 897-901. [9] NIEUWENBURG S A V, MOMMERSTEEG M C, EIKENBOOM E L, et al. Factors associated with the progression of gastric intestinal metaplasia: a multicenter, prospective cohort study[J]. Endosc Int Open, 2021, 9(3): E297-E305. doi: 10.1055/a-1314-6626 [10] ANGERILLI V, VANOLI A, CELIN G, et al. Gastric carcinoma in autoimmune gastritis: a histopathologic and molecular study[J]. Mod Pathol, 2024, 37(6): 100491. DOI: 10.1016/j.modpat.2024.100491. [11] KRIKE P, APPEL M S, SHUMS Z, et al. Autoimmune gastritis serological biomarkers in gastric cancer patients[J]. Eur J Cancer Prev, 2024, 33(1): 29-36. doi: 10.1097/CEJ.0000000000000826 [12] LENTI M V, MICELI E, LAHNER E, et al. Distinguishing features of autoimmune gastritis depending on previous Helicobacter pylori infection or positivity to anti-parietal cell antibodies: results from the autoimmune gastritis Italian network study group (ARIOSO)[J]. Am J Gastroenterol, 2024, 119(12): 2408-2417. doi: 10.14309/ajg.0000000000002948 [13] KITAMURA S, MUGURUMA N, OKAMOTO K, et al. Clinicopathological characteristics of early gastric cancer associated with autoimmune gastritis[J]. JGH Open, 2021, 5(10): 1210-1215. doi: 10.1002/jgh3.12656 [14] CHEN Y, JI X, ZHAO W, et al. A real-world study on the characteristics of autoimmune gastritis: a single-center retrospective cohort in China[J]. Clin Res Hepatol Gastroenterol, 2025, 49(4): 102556. DOI: 10.1016/j.clinre.2025.102556. [15] MICELI E, VANOLI A, LENTI M V, et al. Natural history of autoimmune atrophic gastritis: a prospective, single centre, long-term experience[J]. Aliment Pharmacol Ther, 2019, 50(11-12): 1172-1180. doi: 10.1111/apt.15540 [16] AYESH M H, JADALAH K, AL AWADI E, et al. Association between vitamin B12 level and anti-parietal cells and anti-intrinsic factor antibodies among adult Jordanian patients with Helicobacter pylori infection[J]. Braz J Infect Dis, 2013, 17(6): 629-632. doi: 10.1016/j.bjid.2013.01.009 [17] LI X, FENG M, YUAN G. Clinical efficacy of Weisu granule combined with Weifuchun tablet in the treatment of chronic atrophic gastritis and its effect on serum G-17, PG Ⅰ and PG Ⅱ levels[J]. Am J Transl Res, 2022, 14(1): 275-284. [18] OGUTMEN KOC D, BEKTAS S. Serum pepsinogen levels and OLGA/OLGIM staging in the assessment of atrophic gastritis types[J]. Postgrad Med J, 2022, 98: 441-445. doi: 10.1136/postgradmedj-2020-139183 [19] KISHIKAWA H, NAKAMURA K, OJIRO K, et al. Relevance of pepsinogen, gastrin, and endoscopic atrophy in the diagnosis of autoimmune gastritis[J]. Sci Rep, 2022, 12(1): 4202. DOI: 10.1038/s41598-022-07947-1. [20] CHAPELLE N, MARTIN J, OSMOLA M, et al. Serum pepsinogens can help to discriminate between hpylori-induced and auto-immune atrophic gastritis: results from a prospective multicenter study[J]. Dig Liver Dis, 2023, 55(10): 1345-1351. doi: 10.1016/j.dld.2023.03.015 [21] KRIKE P, SHUMS Z, POŁAKA I, et al. The diagnostic value of anti-parietal cell and intrinsic factor antibodies, pepsinogens, and gastrin-17 in corpus-restricted atrophic gastritis[J]. Diagnostics(Basel), 2022, 12(11): 2784. DOI: 10.3390/diagnostics12112784. [22] 冯真真, 张荣芳, 韩笑笑, 等. 胃蛋白酶原及幽门螺杆菌抗体联合鉴别胃癌和萎缩性胃炎的价值[J]. 医药论坛杂志, 2025, 46(10): 1030-1033, 1038.FENG Z Z, ZHANG R F, HAN X X, et al. The value of combined pepsinogen and helicobacter pylori antibody in differentiating gastric cancer from atrophic gastritis[J]. Journal of Medical Forum, 2020, 46(10): 1030-1033, 1038. [23] ARSHAD U, SARKAR S, ALIPOUR TALESH G, et al. A lack of role for antibodies in regulating Helicobacter pylori colonization and associated gastritis[J]. Helicobacter, 2020, 25(2): e12681. DOI: 10.1111/hel.12681. [24] 李群, 沈君, 史万青, 等. 安徽滁州地区体检人群幽门螺杆菌感染现状及危险因素分析[J]. 中华全科医学, 2025, 23(5): 765-767, 870. doi: 10.16766/j.cnki.issn.1674-4152.003996LI Q, SHEN J, SHI W Q, et al. Analysis of the current situation and risk factors of helicobacter pylori infection in the physical examination population in Chuzhou area, Anhui province[J]. Chinese Journal of General Practice, 2020, 23(5): 765-767, 870. doi: 10.16766/j.cnki.issn.1674-4152.003996 [25] WADA Y, KUSHIMA R, KODAMA M, et al. Histological changes associated with pyloric and pseudopyloric metaplasia after Helicobacter pylori eradication[J]. Virchows Arch, 2020, 477(4): 489-496. doi: 10.1007/s00428-020-02805-9 [26] ANGERILLI V, VANOLI A, CELIN G, et al. Gastric carcinoma in autoimmune gastritis: a histopathologic and molecular study[J]. Mod Pathol, 2024, 37(6): 100491. DOI: 10.1016/j.modpat.2024.100491. -





 下载:
下载: