Relationship between brain symptoms and the expression of intestinal flora metabolites and endoplasmic reticulum stress products in patients with mild carotid stenosis
-
摘要:
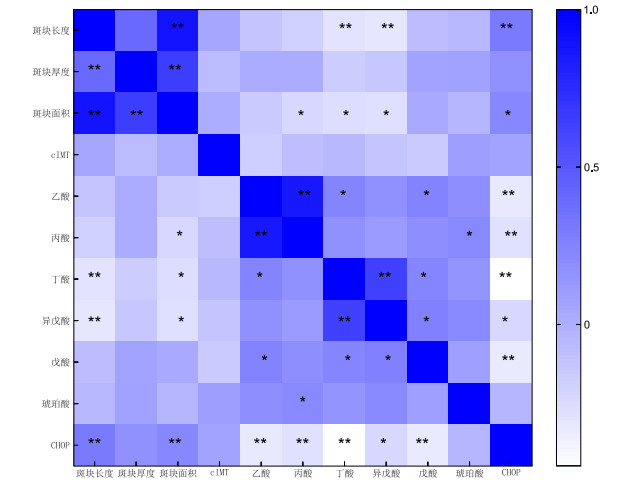
目的 探讨颈动脉轻度狭窄患者脑部症状发生与肠道菌群代谢产物和内质网应激产物表达之间的关系,明确肠道菌群代谢产物介导内质网应激途径在动脉粥样硬化发生发展中的作用。 方法 回顾性分析2021年1月—2023年6月在新疆医科大学第六附属医院接受颈动脉超声检查的87例患者的数据集,采用qRT-PCR分析外周血内质网应激标记物C/EBP同源蛋白(CHOP)mRNA水平。收集患者入院后首次新鲜粪便样本,并通过高效液相色谱法分析样本中短链脂肪酸(SCFAs)水平。 结果 与无症状组相比,症状组高血压例数、斑块长度、斑块面积、外周血CHOP mRNA表达显著增加(P < 0.05),肠道菌群代谢产物乙酸、丙酸、丁酸、异戊酸、戊酸水平显著降低(P < 0.05)。CHOP mRNA表达与斑块长度、斑块面积均呈正相关关系(r=0.299、0.221,P<0.01),与乙酸、丙酸、丁酸、异戊酸、戊酸水平均呈负相关关系(r=-0.324、-0.285、-0.450、-0.233、-0.332,P<0.05)。斑块长度与丁酸、异戊酸均呈负相关关系(r=-0.302、-0.313,P<0.01)。斑块面积与丙酸、丁酸、异戊酸均呈负相关关系(r=-0.230、-0.261、-0.276,P<0.05)。丁酸的预测能力最高,当截止值为8.8时,其AUC为0.902,灵敏度为77.1%,特异度为84.0%。其次为CHOP mRNA表达,当截止值为0.60时,其AUC为0.811,灵敏度为62.9%,特异度为94.0%。 结论 粪便乙酸、丙酸、丁酸、异戊酸、戊酸水平降低以及外周血CHOP mRNA表达增加可能与轻度颈动脉狭窄患者出现脑部症状相关,并且这些变化又与颈动脉斑块特征相关。 Abstract:Objective To explore the relationship between the occurrence of brain symptoms and the expression of intestinal flora metabolites and endoplasmic reticulum stress products in patients with mild carotid stenosis. Methods This study retrospectively analyzed the data sets of 87 patients who received carotid ultrasound examination in our hospital from January 2021 to June 2023, the mRNA level of the endoplasmic reticulum stress marker C/EBP homologous protein (CHOP) in peripheral blood was analyzed by quantitative reverse transcription polymerase chain reaction (qRT-PCR). The fresh stool samples were collected for the first time after admission, and the levels of short-chain fatty acids (SCFAs) in the samples were analyzed by high-performance liquid chromatography. Results Plaque length, plaque area, and CHOP mRNA expression in peripheral blood in the symptomatic group increased significantly (P < 0.05), while the levels of intestinal flora metabolites such as acetic acid, propionic acid, butyric acid, isovaleric acid, and valeric acid decreased significantly (P < 0.05). CHOP mRNA expression was positively correlated with plaque length and plaque area (r=0.299 and 0.221, all P < 0.01), and negatively correlated with the levels of acetic acid, propionic acid, butyric acid, isovaleric acid, and valeric acid (r=-0.324, -0.285, -0.450, -0.233, and -0.332, all P < 0.05). The length of plaque was negatively correlated with butyric acid and isovaleric acid (r=-0.302 and -0.313, both P < 0.01). The plaque area was negatively correlated with propionic acid, butyric acid, and isovaleric acid (r=-0.230, -0.261, and -0.276, all P < 0.05). The prediction ability of butyric acid was the highest. When the cutoff value was 8.8, its AUC was 0.902, sensitivity was 77.1%, and specificity was 84.0%. Followed by CHOP mRNA expression, when the cutoff value was 0.60, its AUC was 0.811, sensitivity was 62.9%, and specificity was 94.0%. Conclusion The decrease of fecal acetic acid, propionic acid, butyric acid, isovaleric acid, and valeric acid levels and the increase of CHOP mRNA expression in peripheral blood may be related to the occurrence of brain symptoms, and these changes are related to the characteristics of carotid plaque. -
表 1 2组接受颈动脉超声检查患者临床特征比较
Table 1. Comparison of clinical characteristics between the two groups of patients who underwent carotid artery ultrasound examination
项目 症状组(n=35) 无症状组(n=52) 统计量 P值 年龄(x±s,岁) 69.60±10.42 69.58±10.70 0.010a 0.992 男性[例(%)] 26(74.3) 39(75.0) 0.006b 0.940 高血压[例(%)] 28(80.0) 28(53.8) 6.239b 0.012 糖尿病[例(%)] 19(54.3) 24(46.2) 0.553b 0.457 吸烟[例(%)] 4(11.4) 6(11.5) 0.000b 0.987 斑块特征 斑块长度[M(P25, P75),mm] 9.00(5.50, 12.00) 6.00(4.00, 9.00) 2.632c 0.008 斑块厚度(x±s,mm) 2.87±1.33 2.53±0.98 1.384a 0.170 斑块面积(x±s,mm2) 35.24±7.07 18.28±2.04 2.305a 0.027 cIMT(x±s,mm) 1.21±0.30 1.19±0.32 0.202a 0.840 DoS(x±s,%) 35.39±11.79 32.28±12.70 1.146a 0.255 肠道菌群代谢产物 乙酸(x±s,μmol/g) 38.97±14.43 52.46±11.58 4.822a <0.001 丙酸(x±s,μmol/g) 26.97±6.03 31.31±5.18 3.585a 0.001 丁酸(x±s,μmol/g) 7.49±1.73 11.77±3.54 6.628a <0.001 异戊酸(x±s,μmol/g) 2.71±0.62 3.61±1.04 4.560a <0.001 戊酸(x±s,μmol/g) 2.76±1.15 3.69±1.50 3.083a 0.003 琥珀酸(x±s,μmol/g) 0.77±0.07 0.79±0.11 0.625a 0.534 内质网应激指标 CHOP mRNA(x±s) 0.57±0.15 0.40±0.16 4.876a <0.001 注:a为t值,b为χ2值,c为Z值。 表 2 CHOP mRNA表达、斑块特征、肠道菌群代谢产物预测脑血管事件的价值
Table 2. The value of chop mrna expression, plaque characteristics and intestinal flora metabolites to predict cerebrovascular events
项目 截止值 AUC 95% CI P值 灵敏度(%) 特异度(%) 斑块面积 21.5 mm2 0.664 0.545~0.783 0.011 62.9 72.0 斑块长度 7.5 mm 0.668 0.549~0.787 0.009 62.9 70.0 CHOP mRNA 0.60 0.811 0.711~0.911 < 0.001 62.9 94.0 乙酸 45.9 μmoL/g 0.765 0.664~0.867 < 0.001 71.4 68.0 丙酸 33.0 μmoL/g 0.697 0.586~0.809 0.002 88.6 46.0 丁酸 8.8 μmoL/g 0.902 0.837~0.967 < 0.001 77.1 84.0 异戊酸 3.3 μmoL/g 0.765 0.665~0.864 < 0.001 85.7 60.0 戊酸 3.4 μmoL/g 0.678 0.564~0.792 0.005 80.0 56.0 -
[1] GABA P, GERSH B J, MULLER J, et al. Evolving concepts of the vulnerable atherosclerotic plaque and the vulnerable patient: implications for patient care and future research[J]. Nat Rev Cardiol, 2023, 20(3): 181-196. doi: 10.1038/s41569-022-00769-8 [2] XIN R J, YANG D D, XU H M, et al. Comparing symptomatic and asymptomatic carotid artery atherosclerosis in patients with bilateral carotid vulnerable plaques using magnetic resonance imaging[J]. Angiology, 2022, 73(2): 104-111. doi: 10.1177/00033197211012531 [3] LI Y, ZHENG S, ZHANG J, et al. Advance ultrasound techniques for the assessment of plaque vulnerability in symptomatic and asymptomatic carotid stenosis: a multimodal ultrasound study[J]. Cardiovasc Diagn Ther, 2021, 11(1): 28. DOI: 10.21037/cdt-20-876. [4] 师晶晶, 鲍婕妤, 张静, 等. 大脑中动脉狭窄粥样硬化斑块特征对因动脉狭窄所致脑梗死的影响[J]. 中华全科医学, 2024, 22(6): 962-965.SHI J J, BAO J Y, ZHANG J, et al. Effect of atherosclerotic plaque characteristics of middle cerebral artery stenosis on cerebral infarction caused by arterial stenosis[J]. Chinese General Medicine, 2024, 22(6): 962-965. [5] COVENEY S, MURPHY S, BELTON O, et al. Inflammatory cytokines, high-sensitivity C-reactive protein, and risk of one-year vascular events, death, and poor functional outcome after stroke and transient ischemic attack[J]. Int J Stroke, 2022, 17(2): 163-171. doi: 10.1177/1747493021995595 [6] 刘莉. 基于菌-肠-脑轴的肠肽与焦虑抑郁关系的研究进展[J]. 中华全科医学, 2022, 20(8): 1388-1391, 1399.LIU L. Research progress on the relationship between intestinal peptide and anxiety and depression based on bacteria-intestine-brain axis[J]. Chinese General Medicine, 2022, 20(8): 1388-1391, 1399. [7] XOURAFA G, KORBMACHER M, RODEN M. Inter-organ crosstalk during development and progression of type 2 diabetes mellitus[J]. Nat Rev Endocrinol, 2024, 20(1): 27-49. doi: 10.1038/s41574-023-00898-1 [8] SÁNCHEZ-ALCOHOLADO L, LABORDA-ILLANES A, OTERO A, et al. Relationships of gut microbiota composition, short-chain fatty acids and polyamines with the pathological response to neoadjuvant radiochemotherapy in colorectal cancer patients[J]. Int J Mol Sci, 2021, 22(17): 9549. DOI: 10.3390/ijms22179549. [9] GHAZNAWI R, VONK J M J, ZWARTBOL M H T, et al. Low-grade carotid artery stenosis is associated with progression of brain atrophy and cognitive decline. The SMART-MR study[J]. J Cereb Blood Flow Metab, 2023, 43(2): 309-318. doi: 10.1177/0271678X221133859 [10] LV H, ZHANG Z, FU B, et al. Characteristics of the gut microbiota of patients with symptomatic carotid atherosclerotic plaques positive for bacterial genetic material[J]. Front Cell Infect Microbiol, 2024, 13: 1296554. DOI: 10.3389/fcimb.2023.1296554. [11] CHEN M, PENG L, ZHANG C, et al. Gut microbiota might mediate the benefits of high-fiber/acetate diet to cardiac hypertrophy mice[J]. J Physiol Biochem, 2023, 79(4): 745-756. doi: 10.1007/s13105-023-00971-3 [12] NOGAL A, VALDES A M, MENNI C. The role of short-chain fatty acids in the interplay between gut microbiota and diet in cardio-metabolic health[J]. Gut microbes, 2021, 13(1): 1-24. [13] TAN C, WU Q, WANG H, et al. Dysbiosis of gut microbiota and short-chain fatty acids in acute ischemic stroke and the subsequent risk for poor functional outcomes[J]. JPEN J Parenter Enteral Nutr, 2021, 45(3): 518-529. doi: 10.1002/jpen.1861 [14] OVERBY H B, FERGUSON J F. Gut microbiota-derived short-chain fatty acids facilitate microbiota: host cross talk and modulate obesity and hypertension[J]. Curr Hypertens Rep, 2021, 23: 1-10. [15] GUO B, ZHANG J, ZHANG W, et al. Gut microbiota-derived short chain fatty acids act as mediators of the gut-brain axis targeting age-related neurodegenerative disorders: a narrative review[J]. Crit Rev Food Sci Nutr, 2023, 27: 1-22. [16] HOSODA K, IMAHORI T, TANAKA K, et al. Contribution of endoplasmic reticulum stress to the clinical instability of carotid plaques in human carotid stenosis[J]. Transl Stroke Res, 2022, 13(3): 420-431. doi: 10.1007/s12975-021-00968-4 [17] PETHÖ D, HENDRIK Z, NAGY A, et al. Heme cytotoxicity is the consequence of endoplasmic reticulum stress in atherosclerotic plaque progression[J]. Sci Rep, 2021, 11(1): 10435. DOI: 10.1038/s41598-021-89713-3. [18] CHE X Y, XIAO Q Q, SONG W, et al. Protective functions of liver X receptor α in established vulnerable plaques: involvement of regulating endoplasmic reticulum-mediated macrophage apoptosis and efferocytosis[J]. J Am Heart Assoc, 2021, 10(10): e018455. DOI: 10.1161/JAHA.120.018455. [19] HONG D, TANG W, LI F, et al. The short-chain fatty acid propionate prevents ox-LDL-induced coronary microvascular dysfunction by alleviating endoplasmic reticulum stress in HCMECs[J]. PLoS One, 2024, 19(5): e0304551. DOI: 10.1371/journal.pone.0304551. [20] KOPCZYŃSKA J, KOWALCZYK M. The potential of short-chain fatty acid epigenetic regulation in chronic low-grade inflammation and obesity[J]. Front Immunol, 2024, 15: 1380476. DOI: 10.3389/fimmu.2024.1380476. -





 下载:
下载: